HUDSON
BIOTECH
Retatrutide (LY3437943) for Clinical-Trial Programs
Source-verified information on retatrutide and the public study record for NCT07467447, together with Hudson Biotech’s positioning as a U.S.-based peptide manufacturing partner for clinical research.
NCT07467447 is publicly listed as a Phase 2 study evaluating once-weekly retatrutide versus placebo in adults with obesity or overweight with weight-related comorbidities, alongside standardized diet and physical activity counseling. Public registry sources reviewed identify Hudson Biotech as sponsor or responsible party.
What Is Retatrutide?
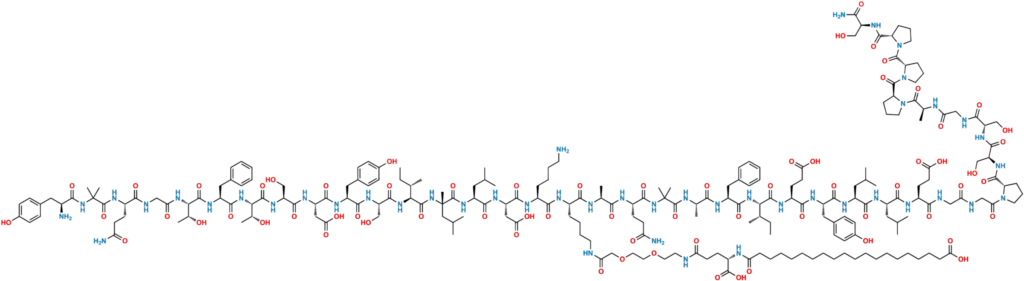
Retatrutide, also called LY3437943, is described in peer-reviewed and official company materials as an investigational triple hormone receptor agonist that activates GIP, GLP-1, and glucagon receptors. Peer-reviewed obesity literature reported substantial body-weight reductions over 48 weeks, and Lilly later reported positive Phase 3 topline results in obesity with knee osteoarthritis in December 2025 and in type 2 diabetes in March 2026. Even with that progress, retatrutide remains investigational, and FDA has not approved it for any condition.
This matters for an SEO page because many users search for retatrutide with mixed intent. Some want scientific context. Some want the exact trial ID. Others are evaluating whether a peptide partner can support legitimate clinical-trial programs. This page is built for those audiences, not for direct-to-consumer sales.

Clinical Trial NCT07467447 Overview
Study Name
Effect of LY3437943 Versus Placebo in Participants Who Have Obesity or Are Overweight (GZBF).
Official Title
Sponsor / Responsible Party
Hudson Biotech
Study Design and Purpose
Public mirrored registry descriptions say the study is interventional, randomized, placebo-controlled, and designed to evaluate once-weekly subcutaneous retatrutide at multiple maintenance dose levels versus placebo, together with standardized diet and physical activity counseling. The primary efficacy evaluation is at Week 24, with additional weight, anthropometric, and safety assessments through Week 48. Estimated enrollment is 300 participants.
Population
The publicly listed population includes adults 18 to 75 years old with obesity, or overweight plus at least one weight-related comorbidity, and without type 2 diabetes.
Study ID
Dates and Public Update
The same mirrored registry pages show:
Start Date: February 15, 2026
Primary Completion Date: March 14, 2027
Study Completion Date: February 17, 2028
ClinicalTrials.gov Last Update: March 12, 2026
Long-Term Follow-Up1
Location
The mirrored location list reviewed shows one current site at Peking University Shenzhen Hospital in Shenzhen, Guangdong, China.
??
Why Choose Hudson Biotech
Hudson Biotech is positioned here as a U.S.-based peptide manufacturing partner for clinical-trial use. On this page, the company should be framed around scientific clarity, trial-focused communication, and documentation-aware execution for investigational peptide programs.
Hudson Biotech supports clinical research organizations, sponsors, hospitals, and procurement teams that need a peptide partner aligned with clinical-trial workflows. Our focus is clear communication, technical rigor, and program support for investigational peptide initiatives.
Peptide Manufacturing for Clinical Trials
Retatrutide (LY3437943)
This section addresses the operational, regulatory, and supply-chain requirements relevant to sponsors, CROs, and clinical-stage programs.
Peptide development and manufacturing for clinical-trial use
Retatrutide (LY3437943) is a synthetic peptide produced via solid-phase peptide synthesis (SPPS), followed by cleavage, purification, and post-synthesis modification such as lipidation to extend half-life.
- Scalable synthesis processes (non-GMP to GMP transition)
- High-purity peptide production suitable for clinical use
- Consistent batch reproducibility aligned with Phase 2–3 trial demands
- Formulation into subcutaneous injectable dosage forms for once-weekly administration
Project coordination for sponsor, CRO, and site-facing teams
Clinical programs for retatrutide involve multi-site, randomized, placebo-controlled trials with long treatment durations and multiple visits.
- Supply timelines aligned with weekly dosing schedules
- CRO integration for protocol adherence and site delivery
- Batch forecasting based on enrollment scale and trial phase
- Cross-functional communication between CMC, clinical operations, and logistics teams
Documentation package support
Clinical-grade peptide programs require full CMC and regulatory documentation to support trial execution and submission readiness.
- Manufacturing process descriptions
- Batch records and traceability documentation
- Certificates of analysis (CoA)
- Stability studies supporting shelf life and storage conditions
- Regulatory-ready modules for clinical submissions
Analytical and release support
Retatrutide requires advanced analytical control to support batch consistency, safety evaluation, and GMP release.
- Purity and impurity profiling (HPLC, LC-MS)
- Peptide identity confirmation and structural verification
- Potency and receptor-binding validation
- Batch release testing under GMP conditions
Placebo or blinded-comparator coordination
Retatrutide trials are typically randomized and placebo-controlled, requiring strict blinding procedures across study arms.
- Matching placebo formulation
- Blinded labeling and packaging
- Comparator management across dosing groups
- Randomization-compatible supply distribution
Storage, shipping, and handling controls
As an injectable peptide biologic, retatrutide requires controlled handling across the full clinical supply chain.
- Cold-chain storage and validated temperature ranges
- Lyophilized or stabilized formulation handling
- Chain-of-custody tracking for clinical sites
- Controlled distribution aligned with dosing schedules
Who We Work With
Hudson Biotech supports organizations involved in peptide development, clinical research, procurement, and manufacturing-related evaluation within lawful institutional and commercial settings.
Hudson Biotech can describe its quality and documentation support with precision once verified. Publish only what can be substantiated, such as quality-system framework, batch record and certificate package, analytical release and impurity-profile support, stability program, labeling or blinding support, and sponsor audit or technical review process.
FAQ:
What is retatrutide (LY3437943)?
Retatrutide, also called LY3437943, is an investigational peptide that activates GIP, GLP-1, and glucagon receptors. Peer-reviewed and official sources describe it as a triple hormone receptor agonist being studied in obesity and type 2 diabetes programs.
What is NCT07467447?
NCT07467447 is the ClinicalTrials.gov identifier for “Effect of LY3437943 Versus Placebo in Participants Who Have Obesity or Are Overweight (GZBF)”, a publicly listed Phase 2 study under Hudson Biotech.
Is retatrutide FDA approved?
No. FDA says retatrutide is not a component of an FDA-approved drug and has not been found safe and effective for any condition.
Can retatrutide be compounded or legally sold online?
FDA says retatrutide cannot be used in compounding under federal law. FDA has also warned about illegal sales of retatrutide products, including products falsely labeled “for research purposes” or “not for human consumption.”
Where is NCT07467447 currently listed?
In the public mirrored registry pages reviewed, one current site is listed: Peking University Shenzhen Hospital in Shenzhen, Guangdong, China. Registry details can change, so the live ClinicalTrials.gov record should be checked before acting.
Who sponsors NCT07467447?
The public study record identifies Hudson Biotech as sponsor, and a public registry snippet says the information was provided by Hudson Biotech as responsible party.
Disclaimer
Investigational-use disclaimer:
Retatrutide (LY3437943) is an investigational molecule. This page is intended for scientific, clinical-trial, and supply-chain audiences.
Medical-information disclaimer:
This page does not provide medical advice, diagnosis, treatment, or dosing instructions.
